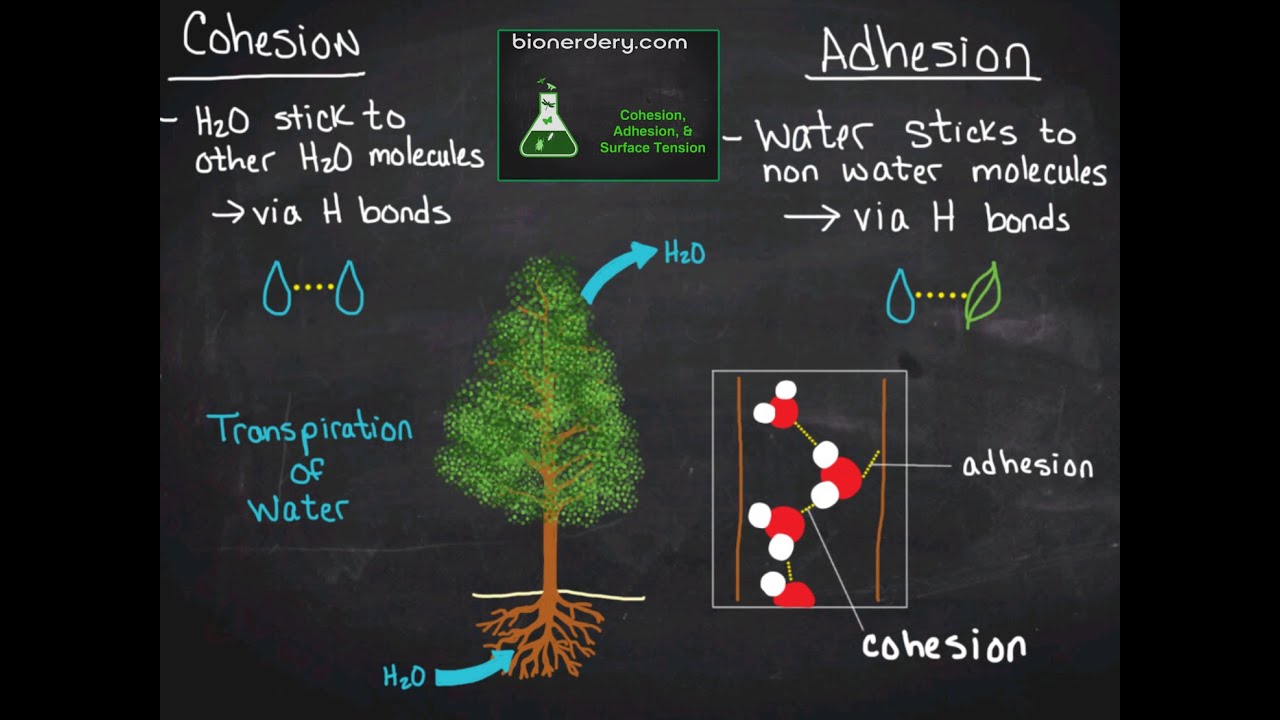
This week in AP Bio we learned about carbon and macromolecules.
Lecture: Carbon
- Carbon is tetravalent
- It makes 4 bonds to get stable which leads to infinite variety
- There is nothing special about life’s building blocks
- Vitalism: belief in life force
- Mechanism: theory that there is nothing special about how life is built
- Isomerism
- Isomers: molecules with the same molecular formula but different structures
- 3 kinds
- Structural: same formula, different order
- cis-trans: same formula, different positioning around double bond
- Enantiomers: same formula, mirror image positioning around a central carbon
- Biological systems tend to use only one of any 2 enantiomeric forms.
Lecture: Macromolecules
- Big! Hence “macro.”
- Made up of a few, common atoms
- Accomplish all life functions
- 4 Main Kinds
- carbohydrates
- lipids
- proteins
- nucleic acids
- Building Macromolecules
- exist in 2 forms
- monomers: simplest
- polymers: a large molecule made of repeating monomers
- exist in 2 forms
- Dehydration synthesis
- Builds more complex molecules
- Water is produced
- Builds Complexity
- Requires energy and enzymes
- Hydrolysis
- Reverse dehydration synthesis
- Lysis: breaking
- Water is needed
- Reduces complexity (Catabolic)
- Releases energy (Exergonic)
- Enzymes required
- Monosaccharides and Disaccharides
- Major carbohydrates used for energy
- Polysaccharides
- Energy Storage
- Amino Acids
- 21 total known
- Every amino acid differs in structure. Structure of the R group varies widely.
- Primary Structures
- the sequence of amino acids in one polypeptide chain
- Secondary Structure
- regular, repeating 3D structures found in all polypeptide chains
- Tertiary Structure
- The specific 3D shape of a particular polypeptide chain aka the “conformation”
- Quartenary Structure
- The specific 3D shape of any protein that is made of more than one polypeptide chain
- Proteins are responsible for all life related cells.
- Hemoglobin: Carries oxygen in red blood cells
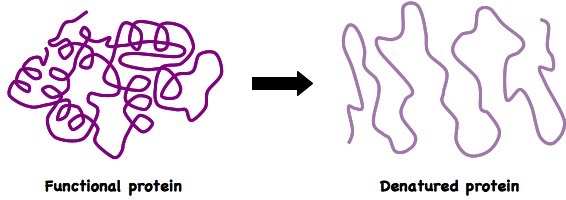
- Denaturation
- change in the structure of a protein

Questions/ Reflections
- I wouldn’t say this week has to relate to any of the big ideas directly, but will help with understanding big idea 2, including “All living systems require constant input of free energy *constant input of energy is necessary for full function of a system.”
Helpful Resources
- Mr. Dunn’s Prezi: https://prezi.com/grlzxhc4gf0c/copy-of-ap-bio-matter-2-macromolecules/